abbott point of care covid test
Personnel and residents who have symptoms of COVID-19 regardless of their vaccination status. The Food and Drug Administration FDA has issued an Emergency Use Authorization for the Abbott ID Now COVID-19 test a molecular point-of-care test that delivers results within minutes allowing healthcare professionals to make clinical decisions during a patient visit.

Trump Announces 750 Million Deal With Abbott Labs For 150 Million Rapid Covid Tests Cbs News
According to Abbott the rapid test which runs on the ID NOW platform is an.

. In this prospective arm of the study the specificity of the Abbott ID NOW test using the kits dry swab was 999 when tested on 1043 prospectively collected paired swabs 95 CI 9947100. ID NOW is an FDA approved CLIA-waived instrument which means that. We offer chronic care management testing in multiple settings.
Food and Drug Administration FDA under Emergency Use Authorization EUA. The ID NOW COVID-19 test is a rapid molecular point-of-care test that detects COVID-19 in 13 minutes or less. A CLIA-certified laboratory or testing site must report all positive SARS-CoV-2 diagnostic and screening test results to the person who was tested or that persons healthcare provider.
The ID NOW COVID-19 assay is now available for use on the ID NOW platform under US. Fifty-five participants were asymptomatic two had previously confirmed COVID-19 and 986 participants were symptomatic with suspected COVID-19 symptoms. Personnel and residents who.
Abbott to market starting next week a fast point-of-care coronavirus test delivering positive results in 5min and negative results in 13min. The COVID-19 pandemic is affecting all of us around the world. Reporting Requirements for Rapid Testing in Point-of-Care Settings.
Abbott received emergency use authorization EUA from the US. Abbotts rapid COVID-19 test isnt the only point-of-care test to receive FDA authorization during the pandemic but Trump has touted it the most by far hailing the speed at which results can. A box containing a 5-minute test for COVID-19 from Abbott Laboratories is pictured during the daily briefing on the novel coronavirus in the.
Major hospital groups in Connecticut opt not to use Abbotts quick turnaround test for. To help provide the critical diagnostic information needed Abbott is currently providing and. The Rhode Island Department of Health RIDOH strongly encourages facilities to test.
Perform moderate high or waived complexity tests. Abbott is putting its resources towards helping you navigate this crisis. This joins Abbotts RealTime SARS-CoV-2 test which was approved under a EUA earlier this month as well as a growing list of companies whose diagnostic tests are being.
The tests can be used in point-of-care settings and at home with an online service provided by eMed. CLIA-certified laboratories or testing sites are no longer required to report negative results for non-NAAT. This study recruited participants presenting for COVID-19 testing at three Melbourne metropolitan hospitals during a period of low COVID-19 prevalence.
Allocation and distribution of instruments and test kits will be determined by Central. What makes this test so different is where it can be used. Abbott has received emergency use authorization EUA from the US.
Food and Drug Administration FDA for the fastest available molecular point-of-care test for the detection of novel coronavirus COVID-19 delivering positive results in as little as five minutes and negative results in 13 minutes. Results from the simple nasal swab are available in 15 minutes through testing individuals suspected of COVID-19. Food and Drug Administration Emergency Use Authorization EUA.
Abbotts BinaxNOW COVID-19 Ag Card test can identify these antigens which are typically detected after symptoms start. Abbott has rapid point-of-care solutions to support your COVID-19 and influenza testing needs. The Abbott PanBio TM COVID-19 Ag point-of-care test was performed alongside RT-PCR.
ABBOTT ID Now COVID-19 POINT OF CARE TESTING 5192020 The following guidance is for institutions that have an Abbott ID NOW instrument and test kits for performing CLIA-waived rapid point of care COVID-19 testing. In addition participants with COVID-19 notified to the Victorian Government were invited to provide additional swabs to. It is used on our ID NOW platform.
Point of care testing to diagnose and manage diabetes and its comorbidities Abbott is transforming care by giving people and their doctors timely information to better manage health. T his test is authorized for use at the Point of Care POC ie in patient care settings operating under a CLIA Certificate of Waiver Certificate. Abbotts molecular point-of-care test for COVID-19 delivers positive results in as little as five minues and negative results in 13 minutes.
The Food and Drug Administration FDA has issued an Emergency Use Authorization for the Abbott ID Now COVID-19 test a molecular point-of-care test that delivers results within minutes allowing healthcare professionals to make clinical decisions during a patient visit. Will deliver 50K testsday to start. The company says it will ramp up its.
Abbott Laboratories ID NOW COVID-19 point-of-care test will be shipped to hospitals care clinics and doctors offices across the country starting Wednesday. Abbott s new point-of-care test for the novel coronavirus that causes COVID-19 was approved by the US. Food and Drug Administration FDA for the ID NOW COVID-19 test in March 2020.
According to Abbott the rapid test which runs on the ID NOW platform is an. Nursing homes and assisted living facilities may use rapid antigen point-of-care POC tests to test personnel residents or visitors for COVID-19. As a leader in diagnostic testing we have a unique responsibility to contribute our expertise to help fight the COVID-19 pandemic.
The BinaxNOW COVID-19 Antigen Self Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 from individuals with or without symptoms or other epidemiological reasons to suspect COVID-19 infection when tested twice over three days with at least 36 hours between tests.
Abbott S Point Of Care Covid 19 Test Detects Coronavirus In As Little As 5 Minutes Biospace
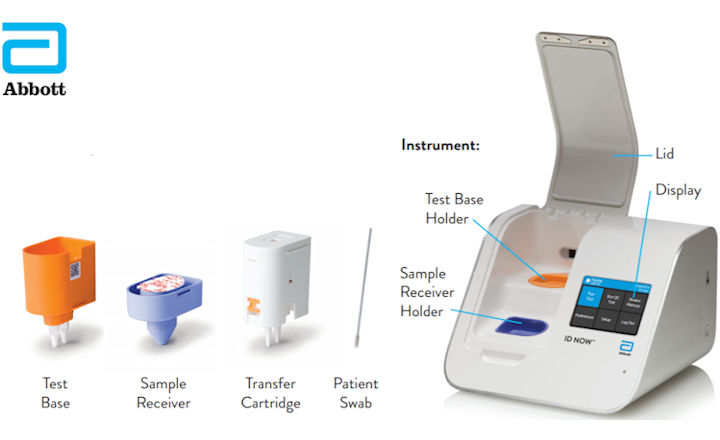
Abbott Id Now Covid 19 Instructions Modified

Scripps Launches Rapid Covid 19 Testing At Hospitals Scripps Health
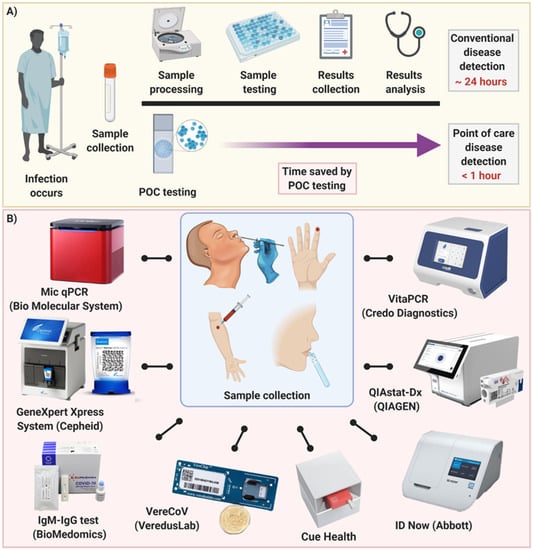
Diagnostics Free Full Text Point Of Care Diagnostics In The Age Of Covid 19 Html
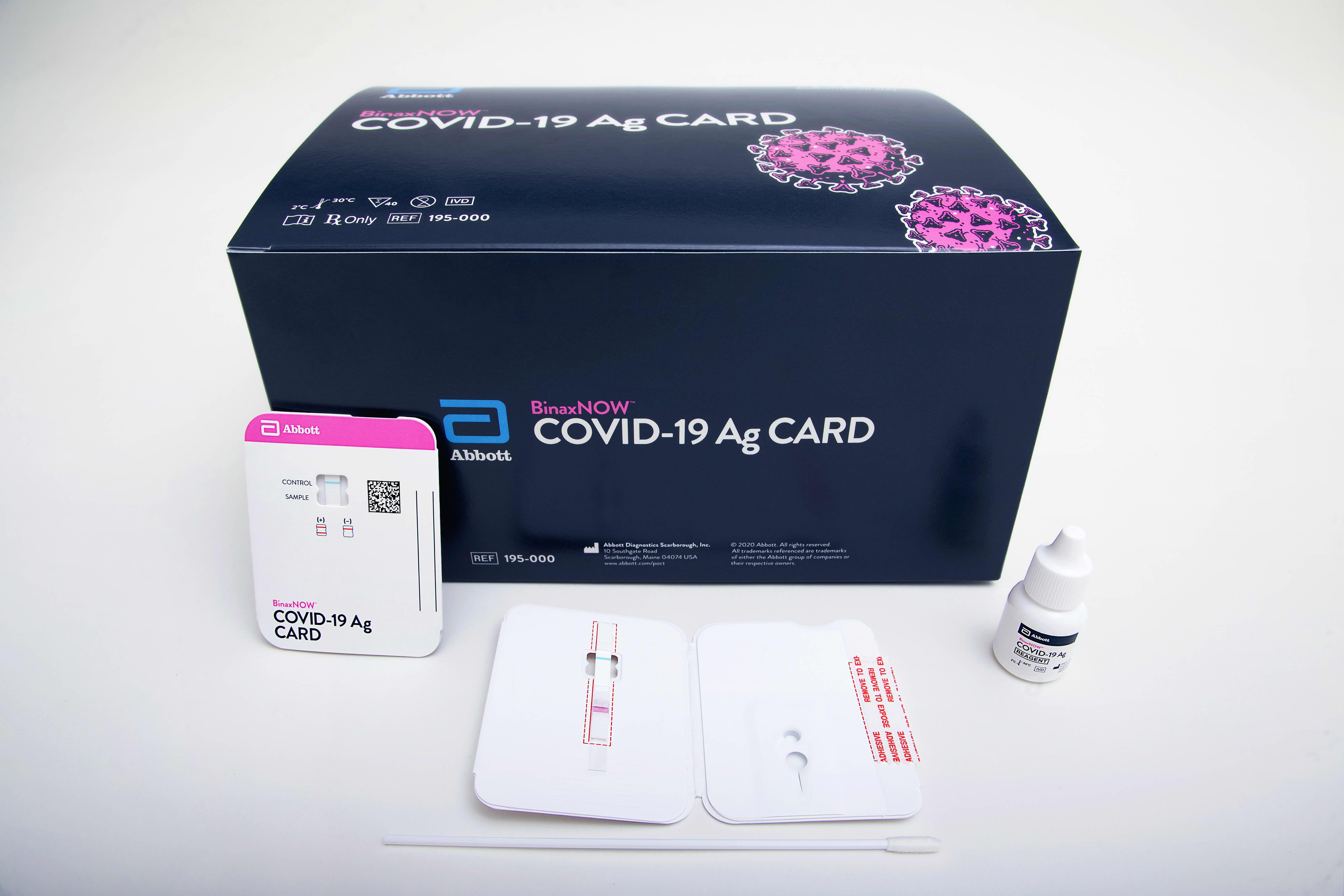
Abbott Labs Rolls Out Rapid Covid Test To Us Schools And Workplaces

Minutes Not Hours Rapid Testing For Coronavirus Youtube

Panbio Covid 19 Ag Rapid Test Device Abbott Point Of Care
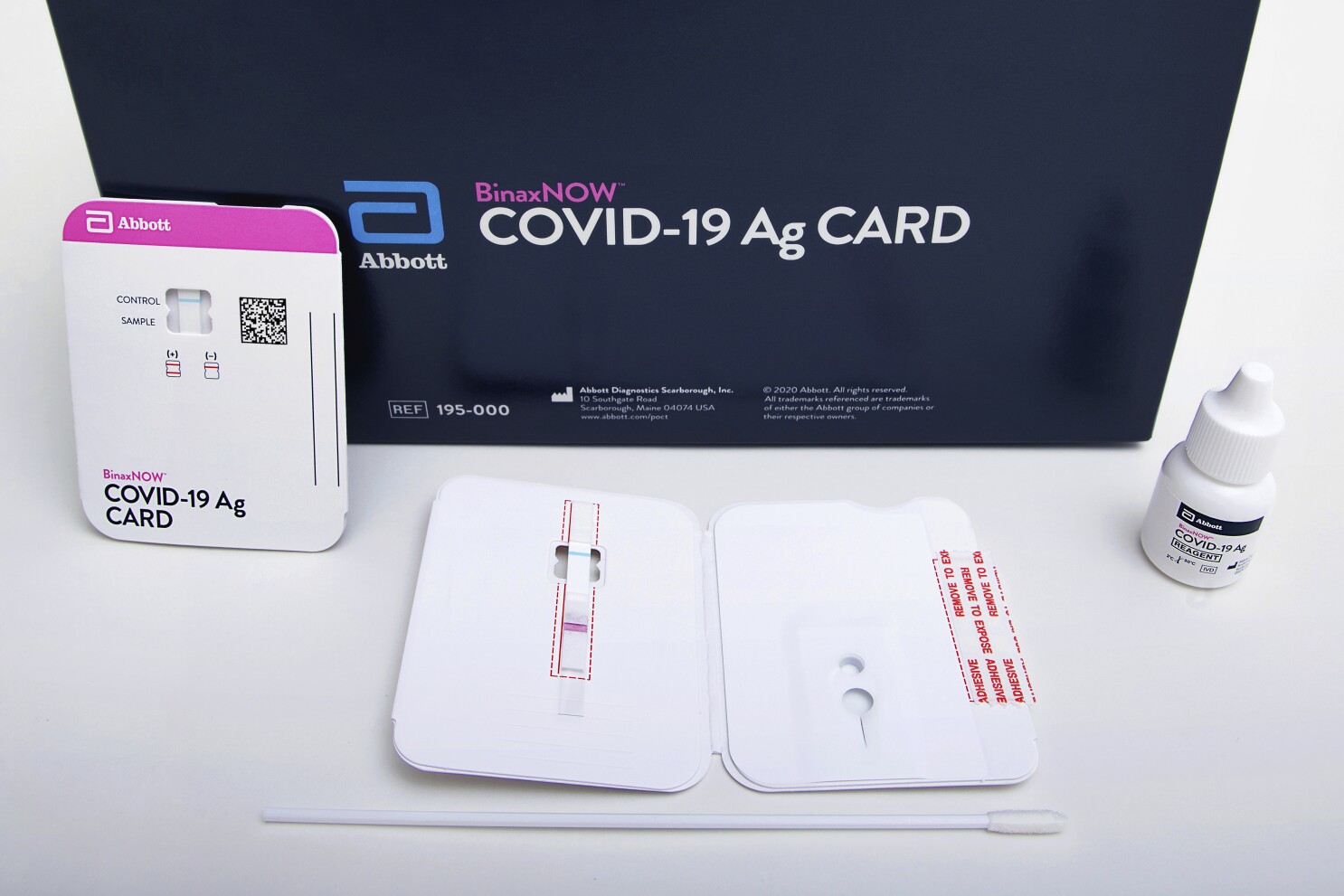
Fda Authorizes Covid 19 Test That Doesn T Need Special Equipment Los Angeles Times

As Problems Grow With Abbott S Fast Covid Test Fda Standards Are Under Fire Kaiser Health News

Steps To Use Id Now Effectively Abbott Newsroom

Our Quick Guide To Rapid Covid 19 Testing Abbott Newsroom
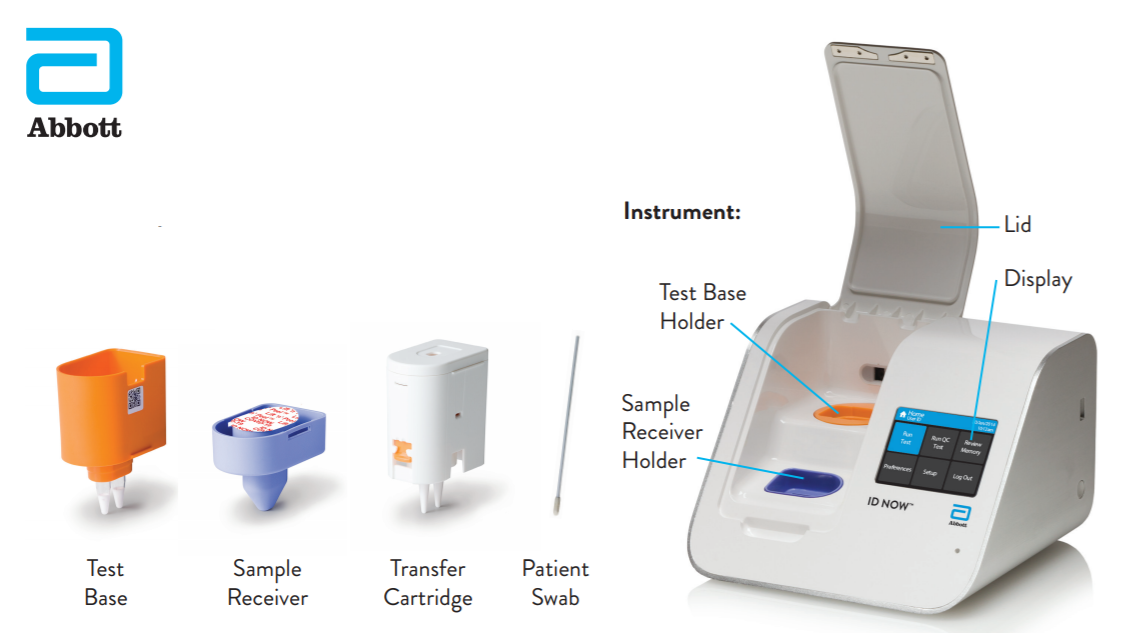
Instant Results From Abbotts Covid 19

Abbott On Twitter We Re Launching A Molecular Point Of Care Test That Delivers Positive Covid 19 Results In As Little As 5 Minutes And Negative Results In 13 Minutes Providing Information Where It Is Needed

Abbott Id Now Covid 19 Detection Test System Us

Rapid Covid 19 Testing Keeping Together Abbott Point Of Care
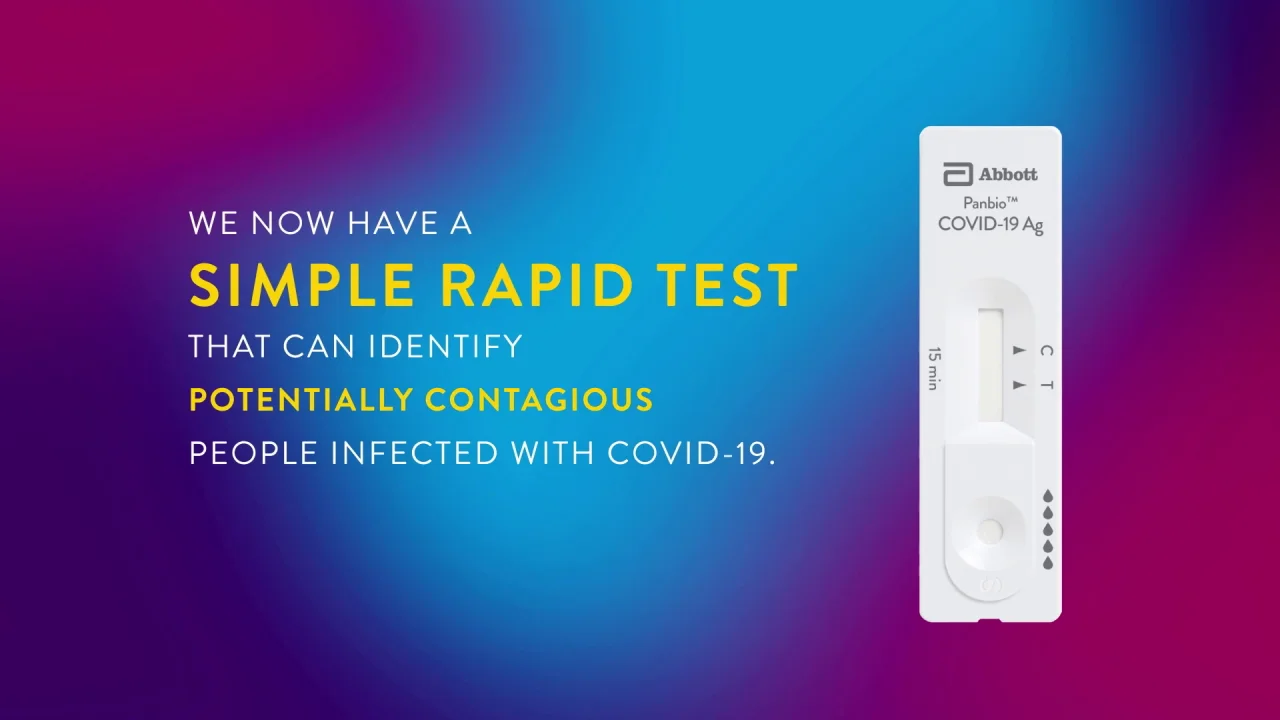
Panbio Covid 19 Ag Test Abbott Point Of Care

Id Now Training Videos Abbott Point Of Care

